Cadmium »
PDB 6w87-7icf »
7d2j »
Cadmium in PDB 7d2j: Crystal Structure of Ixodes Scapularis Glutaminyl Cyclase with A Cd Ion Bound to the Active Site
Enzymatic activity of Crystal Structure of Ixodes Scapularis Glutaminyl Cyclase with A Cd Ion Bound to the Active Site
All present enzymatic activity of Crystal Structure of Ixodes Scapularis Glutaminyl Cyclase with A Cd Ion Bound to the Active Site:
2.3.2.5;
2.3.2.5;
Protein crystallography data
The structure of Crystal Structure of Ixodes Scapularis Glutaminyl Cyclase with A Cd Ion Bound to the Active Site, PDB code: 7d2j
was solved by
K.-F.Huang,
J.-S.Huang,
M.-L.Wu,
W.-L.Hsieh,
A.H.-J.Wang,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 22.98 / 1.60 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 55.127, 71.936, 80.186, 90, 90, 90 |
| R / Rfree (%) | 17 / 19.7 |
Cadmium Binding Sites:
The binding sites of Cadmium atom in the Crystal Structure of Ixodes Scapularis Glutaminyl Cyclase with A Cd Ion Bound to the Active Site
(pdb code 7d2j). This binding sites where shown within
5.0 Angstroms radius around Cadmium atom.
In total only one binding site of Cadmium was determined in the Crystal Structure of Ixodes Scapularis Glutaminyl Cyclase with A Cd Ion Bound to the Active Site, PDB code: 7d2j:
In total only one binding site of Cadmium was determined in the Crystal Structure of Ixodes Scapularis Glutaminyl Cyclase with A Cd Ion Bound to the Active Site, PDB code: 7d2j:
Cadmium binding site 1 out of 1 in 7d2j
Go back to
Cadmium binding site 1 out
of 1 in the Crystal Structure of Ixodes Scapularis Glutaminyl Cyclase with A Cd Ion Bound to the Active Site
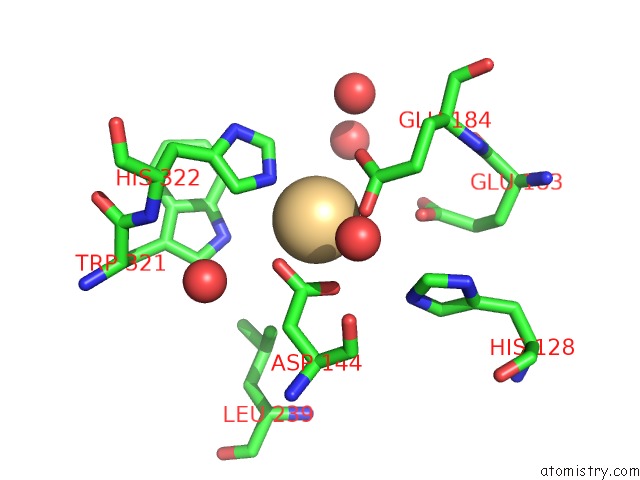
Mono view
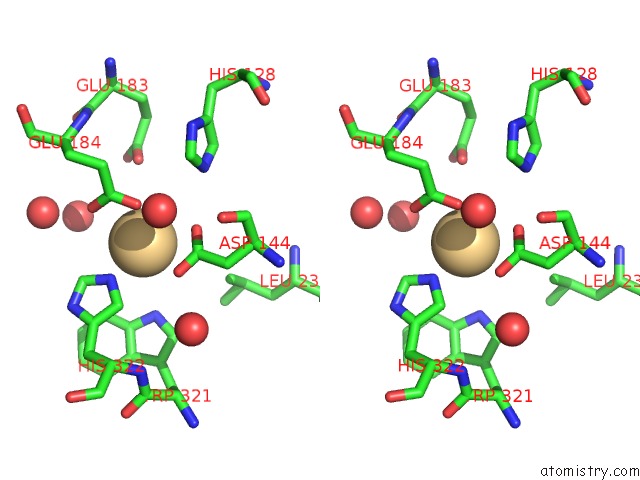
Stereo pair view

Mono view

Stereo pair view
A full contact list of Cadmium with other atoms in the Cd binding
site number 1 of Crystal Structure of Ixodes Scapularis Glutaminyl Cyclase with A Cd Ion Bound to the Active Site within 5.0Å range:
|
Reference:
K.F.Huang,
J.S.Huang,
M.L.Wu,
W.L.Hsieh,
K.C.Hsu,
H.L.Hsu,
T.P.Ko,
A.H-J Wang.
A Unique Carboxylic-Acid Hydrogen-Bond Network (Cahbn) Confers Glutaminyl Cyclase Activity on M28 Family Enzymes. J.Mol.Biol. 66960 2021.
ISSN: ESSN 1089-8638
PubMed: 33774034
DOI: 10.1016/J.JMB.2021.166960
Page generated: Fri Jul 19 19:55:57 2024
ISSN: ESSN 1089-8638
PubMed: 33774034
DOI: 10.1016/J.JMB.2021.166960
Last articles
Zn in 9JYWZn in 9IR4
Zn in 9IR3
Zn in 9GMX
Zn in 9GMW
Zn in 9JEJ
Zn in 9ERF
Zn in 9ERE
Zn in 9EGV
Zn in 9EGW